THEO OVERBEEK CENTER
4TU center on colloid and interface research
4TU center on colloid and interface research

With permission of MIT we have digitized the first 3 of the video lectures of Overbeek
Lecture 1
Lecture 2
Lecture 3

Daily managing directors

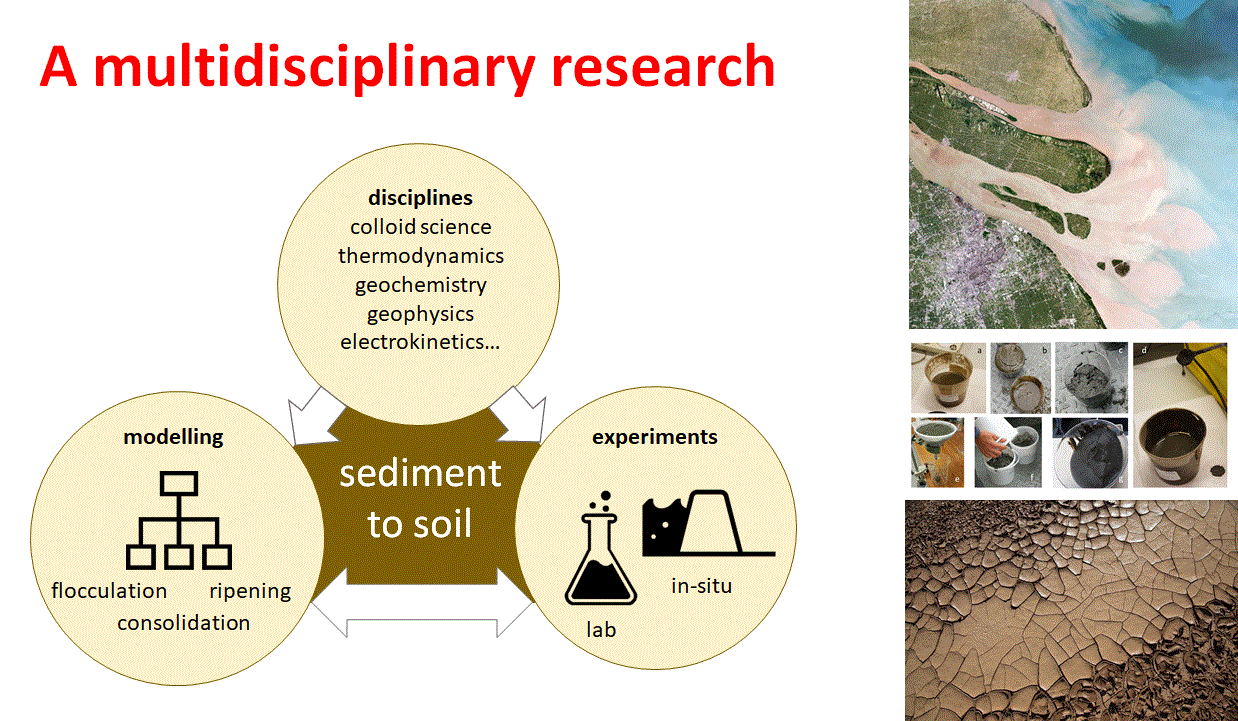
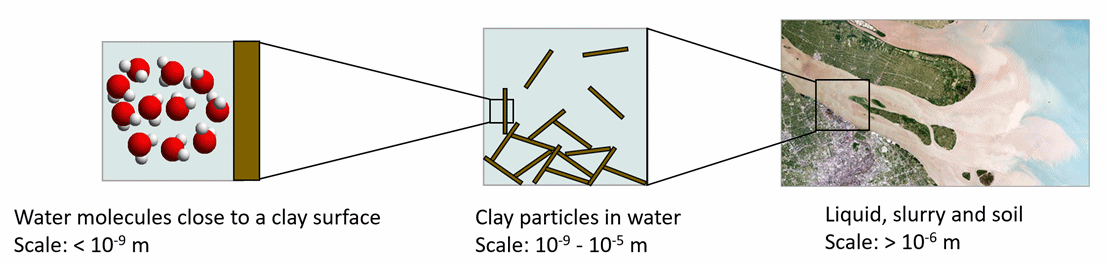
The Theo Overbeek Center (TOC) is a 4TU center was founded with the aim to bring together scientists of different fields of research who study colloids and interfaces. This research not only covers a wide range of disciplines, but also in modelling and experiments, which range from micro-scale to full in-situ. An important research topic within the center is the sendiment-to-soil transition, where the properties of clays at different scales play an important role. Clays can be found all around us and play an important role in many applications, ranging from agriculture to ceramics industry. This 4 TU center wishes to promote multidisciplinary collaboration not only between the 4TU alliance but also between European universities and international universities. This collaboration focuses both on research and teaching. To this end, the center acts as an information hub for scientists and students and a catalyst for research.
The MRI facilities at the Darcy Lab are specially equipped for examining the porosity and composition of materials.
An overview of the NMR scanners ranging from 0.7 to 4.7T can be found here

An overview of the instruments available within MUDNET can be found here
Here an overview is given of articles related to TOC-center.
Here an overview is given of events related to TOC-center.
2021 : Overbeekcenter website was launched
2022 : March improved website
30 January 1911 - 19 February 2007.
Overbeek was born in Groningen. He studied chemistry at the Utrecht University from 1928-1933. After doing his military service and he worked for two years in Belgium. Initially he worked with Jacques Errera at the Université libre de Bruxelles, after that a year with Arend Joan Rutgers at Ghent University.
His doctorate thesis was entitled Theory of Electrophoresis, the Relaxation Effect for which he received a doctorate on May 19, 1941. In his thesis Overbeek's explored the role of the deformation of the electrical double layer surrounding a charged colloidal particle which was set in motion by an external electric field.
After his graduation Overbeek he joined the Philips company. In addition to work his on luminescent screens, he kept looking a the interaction between colloidal particles. This research resulted into the Theory of the Stability of Lyophobic Colloids. This work is still of great influence, and is still used by considerations about the stability of colloids, but also in various other fields where charged surfaces interact with each other. The whole of that theory is now known as DLVO (Derjaguin, Landau, Verwey, Overbeek) theory.
Overbeek became a professor of physical chemistry at the Utrecht University in 1946. During this time he worked on polyelectrolytes,
irreversible thermodynamics, wetting, thin films and biochemical problems. Confronted
with
a challenge, he was in the forefront of recognizing the essentials, defining
the central problem and giving, or helping others to give, the essentials of
the solution. He remained active after his retirement in 1981.
Overbeek received several prestigious awards:
- honorary degrees from Clarkson University and University of Bristol
- the Wolfgang Ostwald Prize.
- honorary membership of the Royal Netherlands Chemical Society
- elected as member of the Royal Netherlands Academy of Arts and Sciences

1911-2007
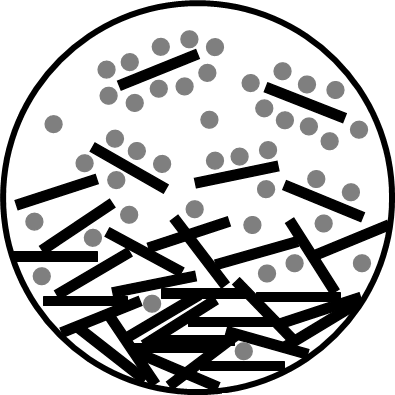
Colloidal systems are systems that have one or more of their characteristic length in the range 10 1000 nm. Suspensions are colloidal when the suspended particles are in the colloidal range. These particles can be solid (like mineral particles) or soft (like polymers or droplets). The suspending medium is a solvent that in colloidal models is assumed to have bulk properties. Porous media can also be seen as colloidal systems when the size of the pores is within the colloidal range. In all cases, the macroscopic properties of the system is governed by its interfacial properties. The behaviour of ions close to charged interfaces drives the electrical properties of the double layer and the fluid transport that results from the forces applied to the system.

The four universities of technology in the Netherlands are jointly committed to strengthening and pooling technical knowledge
4TU contributes to the well-being of the Netherlands by strengthening and pooling knowledge and creativity in the technology sector, and by exploiting it to maximum effect. This is why the four universities of technology (TU Delft, Eindhoven University of Technology, University of Twente and University of Wageningen) are jointly committed to strengthening and pooling their technical knowledge with the aim of producing sufficient numbers of highly qualified engineers and technical designers, of conducting outstanding and socially relevant research of an international standard, and of promoting cooperation between research institutes and businesses
The Theo Overbeek Center is a cooperation between these 4 universities on colloid and interface research
The 4 universities which are :





As a Theo Overbeek Center we are actively seeking cooperation both national and international
An important aspect of the Theo Overbeek Center is the support of teaching. Indeed, Prof Overbeek was one of the first to recognize the power of new multi media. In 1971 he started videotaping his course: a 55-lecture series about colloid and surface chemistry. There was already a publication in Chemical Engineering News in 1971 entitled 'MIT videotape series can replace textbooks' [1]. In a report in 1973 Overbeek himself refers to his videotaped lectures as made during his stay at MIT in the USA indicating that students so always would have access to his course. In the preface of his report of 1973 he writes:


With permission of MIT we have digitized the first 3 parts of the video lectures of Overbeek
Ending the preface with:
Short clip of one of the videos Overbeek made in this course on for MIT where he introduces himself
>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>
Dr. Claire Chassagne / Dr.ir. Leo Pel
Email: Overbeekcenter@outlook.com
Alternatively email for information: C.Chassagne-at-tudelft.nl or l.pel-at-tue.nl
Links
MUDNET (Netherlands)
Darcy-center (Porous media science and technology)
IRCCM
- The International
Research Centre for Clay Micromechanics
Porelab (Norway)
Laboratoire Navier (Paris)
Research Unit of Bio-resource Colloid Engineering (Tsukuba, Japan)





Powered by Silex website builder
Contact email: Overbeekcenter@outlook.com
Design: Leo Pel, Claire Chassagne