The effect of ferrocyanide ions on sodium chloride
crystallization in salt mixtures
Introduction
The use of crystallization inhibitors has been proposed as a
potential preventive treatment method against damage and is
extensively tested for crystallization of single salts. However, in
practice always mixtures of salts are present. Therefore, before
using inhibitors in practice there is a strong need to explore their
effect on salt mixtures. In this research, we studied the effect of
ferrocyanide ions on NaCl crystallization in salt mixtures of NaCl -
LiCl. In this study, a specially designed Nuclear Magnetic Resonance
(NMR) set-up was used (see fig 1). NMR is used for carrying out
non-destructive, quantitative and simultaneous measurements of
hydrogen (H), sodium (Na) and lithium (Li) ions in the droplet To
obtain visual information time lapse microscopy of the
crystallization was performed using a Dino-lite© digital microscope.
Four LED’s were placed at the bottom of the sample holder to provide
additional lighting within the enclosed NMR set-up. The capture of
photomicrographs along with the NMR measurements gives the
possibility to visualize the drying droplet while simultaneously
obtaining information about the amount of dissolved salt ions.
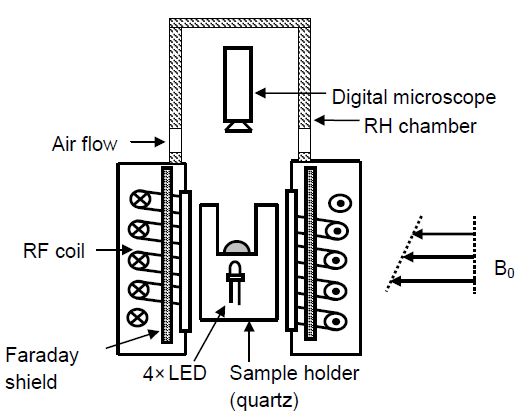 Fig 1 Schematic diagram of NMR set-up used for droplet
drying experiments. A 300 μl droplet was placed on a quartz
glass holder.
Fig 1 Schematic diagram of NMR set-up used for droplet
drying experiments. A 300 μl droplet was placed on a quartz
glass holder.
Time lapse microscopy of the drying droplet was performed
by incorporating a digital microscope in the set-up. Using a
Perspex chamber
the humidity around the droplet was controlled.
Paradoxical drying
The ternary phase diagram of a NaCl-LiCl-H2O mixture at 25
oC is given in Fig. 6.7, where the LiCl concentration (m) is
plotted as a function of the NaCl concentration [85]. The
solubility equilibrium line is shown by a red line. Everywhere
on this line NaCl will crystallize and LiCl will precipitate
only at very high concentrations, i.e., 19.84 m LiCl and 0.03m
NaCl. As an example, the expected paths during evaporation of a
solution are shown by the solid lines A, B and C for three
different initial ion concentrations
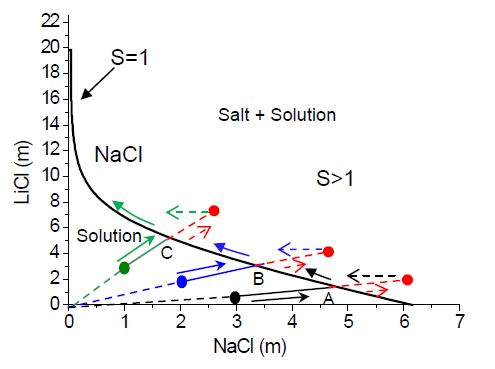
Fig 2.The ternary
phase diagram of NaCl-LiCl-H2O at 25oC. During evaporation the
paths A, B and C (shown by solid black, blue and green lines)
will be followed for salt mixture of compositions 3m NaCl - 1m
LiCl, 2m NaCl - 2m LiCl, and 1m NaCl - 3m LiCl, respectively. If
no crystal appears at the intersection of path A, B, C and the
equilibrium line (S = 1), the concentration will keep on
increasing (supersaturation), as indicated by dotted red arrows.
After crystallization the system will return to the equilibrium
solubility line.
Three different salt ion concentrations were tested, i.e.,
3m NaCl - 1m KCl, 2m NaCl - 2m LiCl, and 1m NaCl - 3m KCl. For
all these experiments, 0.01m inhibitor concentration was
used. The results are shown in Fig. 3. The solid lines are the
expected paths to be followed upon drying of the droplet. As can
be seen from the figure, the experimental data points coincide
very well with the expected paths. The salt ion concentration
first increases to the equilibrium line. At this point a salt
crystal appears, as was confirmed from the pictures taken by the
digital microscope. After the onset of crystallization the
concentration of NaCl will decrease and the equilibrium
solubility line is followed, as shown by dotted arrows. These
results show that multi-nuclear NMR is a powerful experimental
tool to validate the phase diagrams. For the mixture of NaCl -
LiCl also no significantly higher NaCl supersaturation was
observed in the presence of inhibitor. These results indicate
that in the presence of another salt, ferrocyanide ions are not
able to highly supersaturate NaCl.
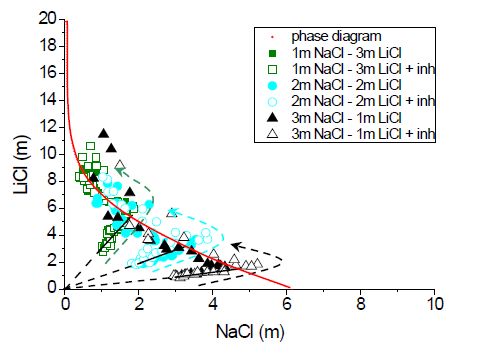 Fig 3
Fig 3:
The three tested concentrations of NaCl - LiCl
salt mixtures shown in the ternary
phase diagram of the NaCl - LiCl - H2O system. The open
symbols denote salt mixtures with
inhibitor and the closed symbols denote salt mixtures
without inhibitor. The solid lines show
the expected pathways to be followed before the equilibrium
line is reached.
However, a clear habit modification was also seen in this case. The
pictures showing the crystal morphology at the end of the
experiments are shown in Fig. 4, showing the crystal morphology in
case of the mixture of NaCl - LiCl without and with inhibitor,
respectively. As can be seen in more spreading occurred and
dendritic crystals were formed in the presence of inhibitor.
 Fig 4
Fig 4:
Images showing the crystal morphology at the end
of the drying experiment for
(a) LiCl and (b) LiCl + 0.01m inhibitor.
Effect of inhibitor
In the absence of inhibitor after approx. 15 hours, the
saturation concentration was achieved in the top few mm of the
sample . This causes a dramatic drop in the drying rate for
salt saturated brick. Since, most of the salt crystallized as
sub-florescence, it causes a more severe blockage of the pores
near the drying surface. However, in the presence of inhibitor
the crystal morphology changes from cubic to dendritic.
The salt solution creeps along the branches of the
dendrites and transports more and more dissolved salt
ions towards the drying surface causing the
efflorescence observed at the end of drying experiment.
Pictures of the materials with efflorescence are shown in fig.
4. Approx. 26% and 69% of the salt crystallized as
efflorescence in the presence of 0.001 m and 0.01 m inhibitor
respectively. Because of the formation of efflorescence
in the presence of inhibitor the average salt ion
concentration inside the brick remained below saturation.
Therefore, the system remained open and less blockage occurred
compared to the salt saturated system without inhibitor. As a
consequence of this no dramatic drop in drying rate was seen
and the paradoxical disappears.
Conclusion
For salt mixtures it is found that
the inhibitor will result in a much lower supersaturation than
that of the single salt. For both single salt and salt mixtures,
the crystal morphology changes completely from bigger cubic
crystals to dendritic crystals in the presence of inhibitor. The
crystal size decreases significantly at high inhibitor
concentrations.
- An extensive description can be found in:
Sonia Gupta, Micheal
Steiger, Leo Pel, Klaas Kopinga, The effect of ferrocyanide
ions on sodium chloride crystallization in salt
mixtures, submitted Crystal
Growth and Design (2014)
S.Gupta, Sodium
chloride crystallization in drying porous media: influence of
inhibitor, Ph.D. thesis, Eindhoven University of Technology (2013)
(Download
2.6 Mb)
 Back
to main page
Back
to main page



