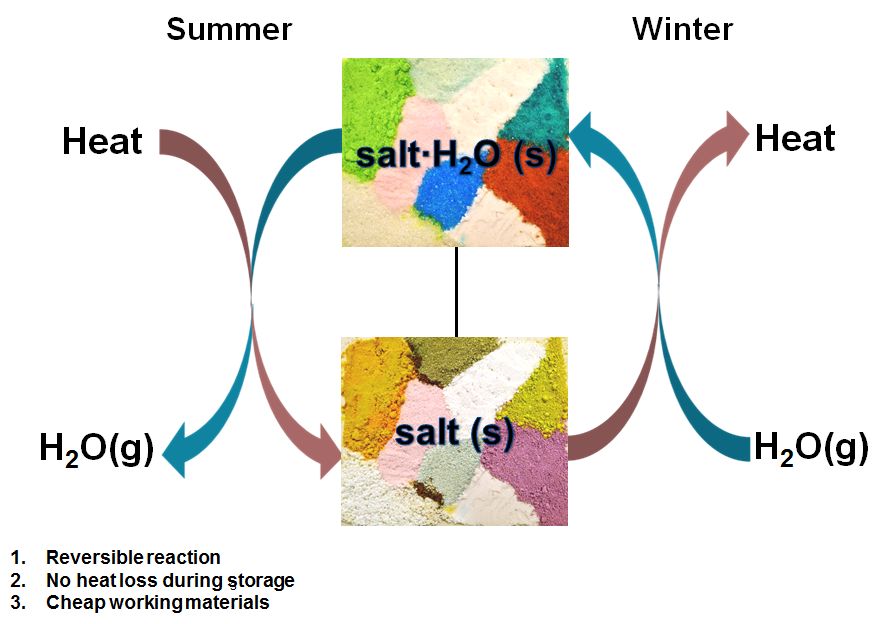
Fig 1 Schematic diagram of the loading and unloading by hydration and dehydration of a salc crystal in order to store heat

P.A.J. Donkers, Experimental study on thermochemical heat storage materials, Ph.D. thesis, Eindhoven University of Technology, the Netherlands (2015), PhD-Donkers-2015 3.5 Mb.pdf
P.A.J. Donkers, L. Pel, O.C.G. Adan, Hydration / dehydration cycles of salt hydrates – studied with NMR–, Conference Proceeding EuroSUn 2014, Aix-les-Bains (France), 16 - 19 September 2014, http://proceedings.ises.org/paper/0154-Donkers.pdf
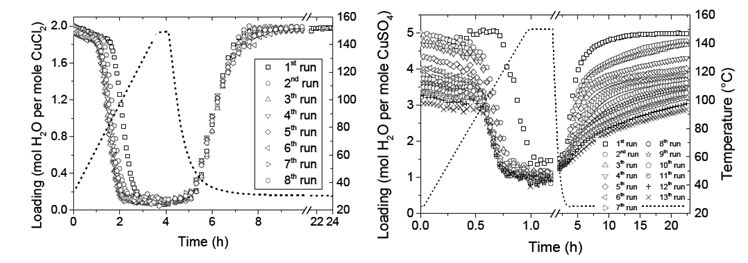
P.A.J. Donkers, K. Linnow, L. Pel, M. Steiger, O.C.G. Adan, Na2SO4•10H2O dehydration in view of thermal storage, Chemical Engineering Science 134, 360–366 (2015)
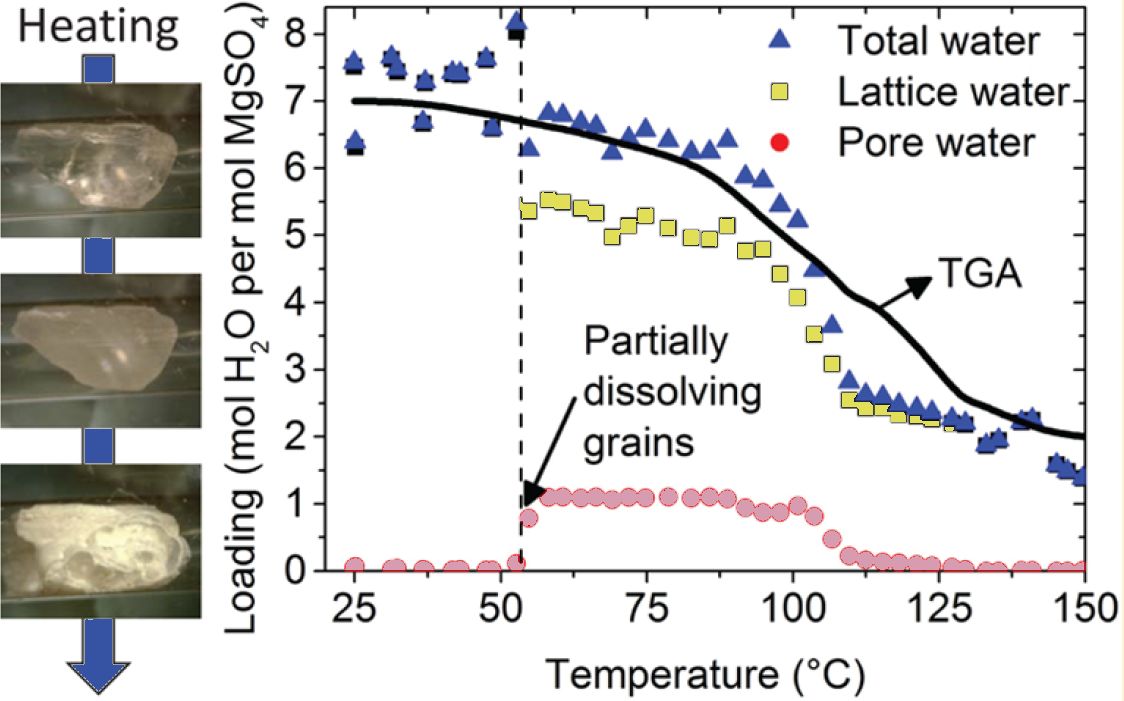
Pim A.J. Donkers, Steffen Beckert, Leo Pel, Frank Stallmach, Michael Steiger, and Olaf C.G. Adan, Water Transport in MgSO4·7H2O During dehydration in view of thermal storage, J. Phys. Chem. C119 28711–28720 (2015)
P.A.J. Donkers, L. Pel, O.C.G. Adan, Experimental studies for the cyclability of salt hydrates for thermochemical heat storage, Journal of Energy Storage 5 25–32 (2016)
P.A.J. Donkers, L. Pel, L.,M. Steiger, O.C.G. Adan, O.C.G. Deammoniation and ammoniation processes with ammonia complexes. AIMS Energy, 4(6), 936-950 (2016).
Pim Donkers, Leo Pel, Dehydration/hydration of granular beds for thermal storage applications: a combined NMR and temperature study, Int. J. of Heat and Mass Transfer 105 826–830 (2017)