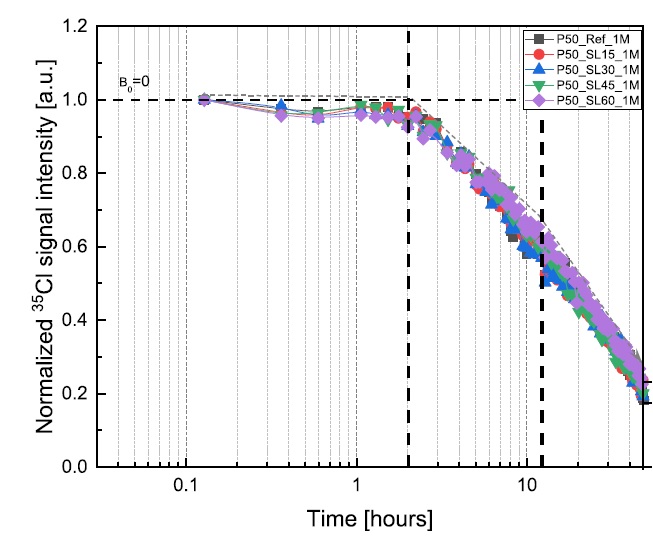

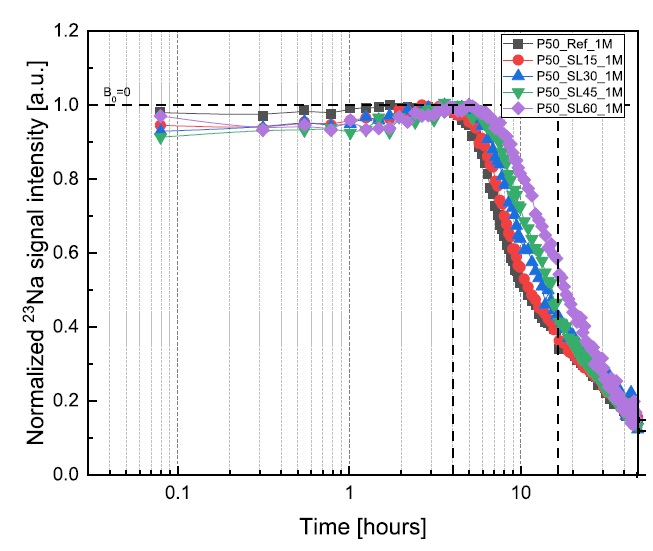
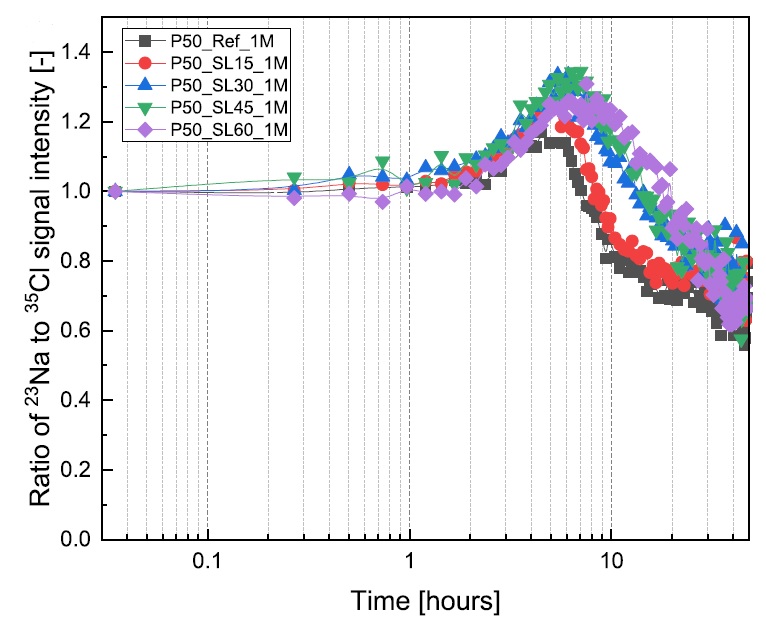
Yanliang Ji, Leo Pel, Xiaoxiao Zhang, Zhenping Sun, Cl- and Na+ ions binding in slag and fly ash cement paste during early hydration as studied by 1H, 35Cl and 23Na NMR, Const. Build. Mat. 266B,121606 (2021).
Yanliang Ji, Leo Pel, Zhenping Sun, NMR study on the early-age hydration and ion binding of the cement paste prepared with NaCl solutions, Cement and Concrete Composites 129, 104489 (2022).